About us
The project is focused on the development of new anti-cancer agents that selectively counteract malignancies.
Research Objectives:
- To design, prepare and test effective anti-cancer agents, especially with regard to the selective killing of malignant cells.
- To analyse and understand the molecular biology and the resistance of cancer stem cells towards apoptosis.
- To propose and verify effective therapeutic strategies which can be used in clinical medicine to minimize cancer relapses.
In collaboration with Australian research group (Mitochondria, Apoptosis and Cancer Research Group, Griffith University, Southport, Qld, Australia), the laboratory focuses on the design and development of novel anti-cancer agents, in particular vitamin E analogues, that are efficient and selective for malignant cells. The researchers are especially interested in the molecular mechanism of apoptosis caused by such agents, which includes pathways of mitochondrial destabilisation. They have defined and characterized the group of ‘mitocans’, small molecules relaying their anti-cancer activity via mitochondria. These agents are particularly intriguing from the translational point of view, since it is now clear that tumours are extremely heterogeneous in their mutations, even within different regions of the same tumour. Therefore, mitochondria may present an invariant target that can be utilized for cancer therapy.
A particular focus of the group is on mitocans from the group of vitamin E analogues, represented by the redox-silent compound α-tocopheryl succinate (α-TOS). This agent exerts high apoptogenic activity towards a variety of cancer cell lines while being non-toxic to normal cells. This in vitro pro-apoptotic activity is matched by its anti-cancer activity in mouse models of tumours, which the group has shown for colorectal, breast and mesothelioma tumours. This finding endows α-TOS with substantial translational spin and/or makes it a lead compound for the design of more efficient agents.
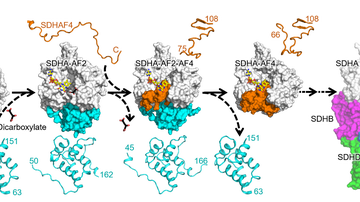
In the attempt to characterize the pathway(s) regulating apoptotic signalling in cancer cells triggered by α-TOS, the researchers discovered a new target of anti-cancer drugs, the mitochondrial complex II (CII). Interestingly, they found that α-TOS (and similar compounds) interacts with the proximal and distal ubiquinone (UbQ)-binding site of CII, thereby replacing UbQ in CII. CII, by means of its succinate dehydrogenase (SDH) activity, converts succinate to fumarate as a component of the tricarboxylic acid (TCA) cycle. Electrons released from this reaction are normally intercepted by UbQ and donated to CIII of the electron transport chain (ETC), part of oxidative phosphorylation. In case UbQ is replaced by α-TOS or similar compounds, the electrons recombine with molecular oxygen and cause generation of superoxide, triggering the apoptosis signalling cascade. Since genes coding for the subunit of CII mutate only exceptionally, the complex presents a very intriguing target for anti-cancer drugs.
In the attempt to increase the apoptogenic activity of α-TOS, the agent was modified by tagging with the cationic triphenylphosphonium (TPP) group, which causes the accumulation of the agent (mitochondrially targeted vitamin E succinate, MitoVES) at the interface of the mitochondrial inner membrane and the mitochondrial matrix, enhancing the apoptogenic activity of the parental compound by 1–2 orders of magnitude. Importantly, this increase in toxicity towards cancer cells did not compromise the selectivity of the agent for malignant cells. The approach of the group to cancer therapy by tagging anti-cancer agents with lipophilic cations epitomised by TPP presents a novel paradigm of efficient cancer therapy. The group is further pursuing this line of research to synthesize novel, more efficient anti-cancer agents targeting mitochondria. In particular, they plan to initiate translational research, whereby delivering their novel agents to the clinic. If successful, this may change the landscape of current and future cancer therapeutic modalities potentially leading to efficient cancer treatment.